GD-MS Analysis and Thermal Upgrading
An approximately 250 g sample of the +65 mesh (>210 micron) Miller flotation concentrate produced by SGS Canada Inc. (Lakefield) as part of a bench-scale flotation optimization program was submitted to Evans Analytical Group, Liverpool NY (EAG-NY), where it was prepared for chemical survey analysis by GD-MS. In this method, the sample (graphite) is mounted as the cathode in the glow discharge cell and analyzed directly. Argon is generally used as the discharge (plasma) gas, which atomizes the sample. Atomized species diffuse into the discharge plasma, where they are ionized. Carbon ions together with analyte ions (deemed impurities, here) are then extracted from the cell and accelerated into the mass analyzer for detection. The plasma atomization process continues until a sufficient mass of the sample has been atomized, to ensure that the acquired results are representative of the analyzed sample. The mass spectrometer determines the ion beam ratios of analyte ions versus carbon ions, thereby identifying the elements present in the sample, and their mass fractions. Reference sample analysis is used to determine the relative sensitivity factors for each element, which then determines the limits of detection for each element.
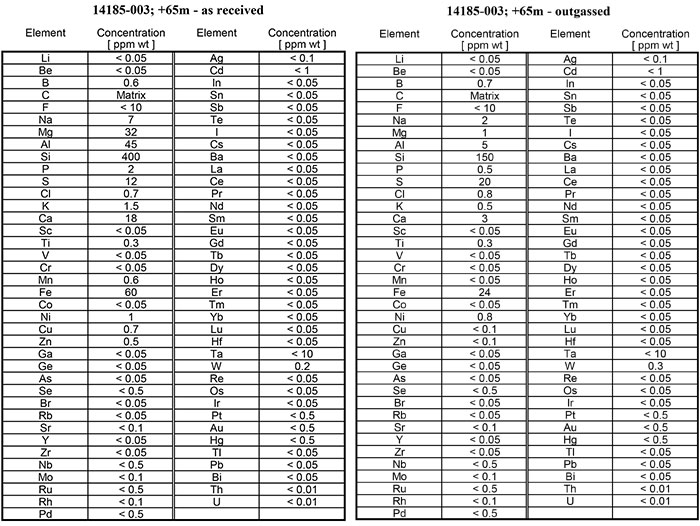
For the assay results reported here, larger aliquots were taken so that the assay protocol could be optimized for fast flow GD-MS measurements, resulting in higher instrumental sensitivity. Approximately 5g samples of the Miller graphite were pressed into self-supporting wafers without a binder. These samples were then assayed by FF-GDMS "as received", and also after Rapid Thermal Upgrading (helium outgassing).
For the sample reported here, total graphite purity was calculated by difference, both before and after brief thermal upgrading (by helium outgassing) of the approximately 250 g sample of Miller hydrothermal graphite flotation concentrate. This sample yielded grades and Equivalent Boron Content (EBC) values, as follows: "As received": 99.39% C(t), EBC 1.26 ppm; "helium outgassed": 99.74% C(t), EBC 1.34 ppm.
C(t) % was calculated as "100% minus (sum of all impurity concentrations (%))". In those instances in which the GD-MS assay reported an element concentration as being below the detection limit for this method, the detection limit concentration was used in the calculation of total impurity content. For example, in all assays, Cadmium was reported as "<1 ppm" concentration, by weight. For the purposes of the purity calculation, Cadmium = 1ppm was used, as it is the most conservative approach to interpreting the data. Therefore, the calculated graphite purity values reported here are the minimum possible values which can be derived from the data.
The impurity concentrations obtained by GDMS were used to calculate the Equivalent Boron Content (EBC) of the graphite, as defined in ASTM Method C1233-09, "Standard Practice for Determining Equivalent Boron Contents of Nuclear Materials", in conjunction with ASTM Standard D7219-08, "Standard Specification for Isotropic and Near-isotropic Nuclear Graphites", which lists the 16 elements of concern with respect to the EBC criterion. EBC is a means of estimating the potential for the impurities contained in the graphite to absorb neutrons when exposed to the controlled neutron flux within a nuclear reactor. Any impurities absorbing neutrons would adversely affect the rate and the control of the nuclear chain reaction.
EBC is calculated as the sum of the EBC of each impurity, such that EBC (impurity) is equal to (EBC factor for impurity) multiplied by (concentration of impurity (ppm)). Each EBC factor was obtained from Table 1 of ASTM Method C1233-09. A number of contaminants of concern were below the detection limit of the GDMS assay procedure, so the concentration associated with each respective detection limit was submitted for the calculation of the EBC of those contaminants, in accordance with the method.
Since October 2013, all samples submitted to Evans Analytical Group for laboratory characterization tests have been analyzed by GD-MS. When interpreted using the ASTM methods described above, every sample has returned calculated EBC values between 1 and 3 ppm boron equivalent.
Rapid Thermal Upgrading (RTU) is a method for quickly eliminating heat-labile impurities from a graphite sample, by exposing the sample to high heat in the presence of an inert gas atmosphere. Work is proceeding to scale up the thermal upgrading procedure, in order to make available larger samples of high-purity graphite without exposing it to harsh chemicals (caustic/acid).
The results of RTU, reported here, show that the total impurity concentration had declined from 609 ppm to 236 ppm, after a 3 minute heat treatment, representing a reduction of 61%.
The Company is working to scale up the RTU process from the bench-scale procedures currently in use.
Laboratory Facilities and Procedures
Elemental analysis by GD-MS were conducted by Evans Analytical Group, Liverpool NY. Rapid Thermal Upgrading (RTU; helium outgassing) was conducted at the same facility. Treatment conditions were: flowing helium atmosphere, temperature 2300 C, duration 3 minutes.